 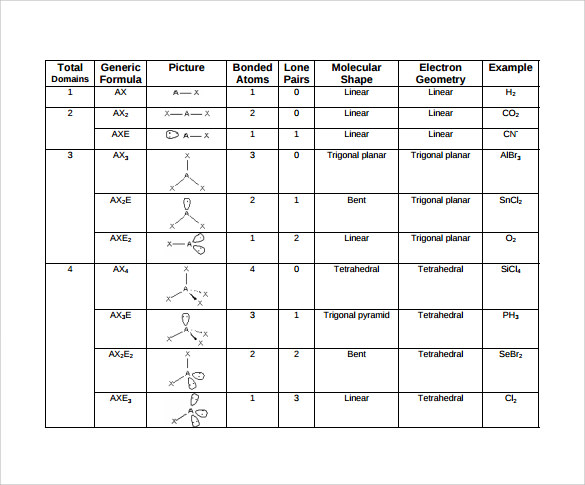
Lone pairs are non-bonded valence electrons and have more repulsions than bond pairs. The bond pairs find a position in space to minimize repulsive interactions and increase the distance between them due to electron repulsion. The repulsive interactions between the valance electrons of the atoms are the basis for this theory. Sidgwick and Powell’s VSEPR theory provides a basic method for predicting the form of a covalent molecule. Let me give you a brief description of the VSEPR Theory There are also two bond pairs, one between oxygen and silicon and the other between silicon and oxygen.Īs a result, there are no lone pairs and two bond pairs.Īccording to the VSEPR theory, if a molecule has Sp hybridization and two bond pairs, but no lone pair on the central atom, the molecule’s molecular and electron geometry is linear. 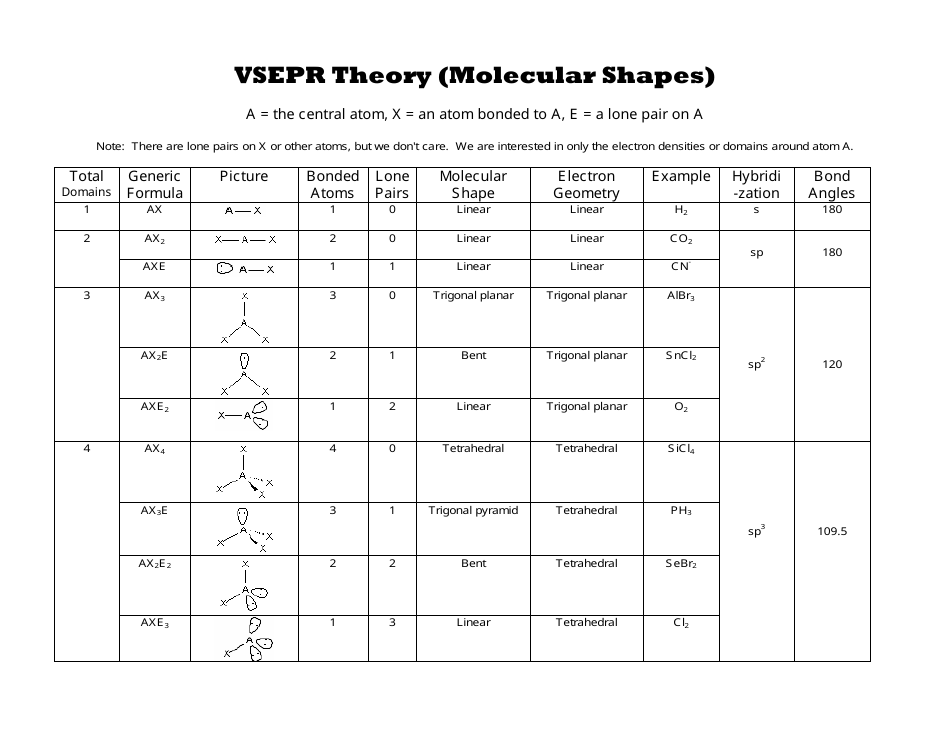
There are no lone pairs of electrons on the central atom of the Silicon dioxide Lewis structure. It has no lone pair on it but has two atoms attached to it.ĭetermine the number of bond pairs and lone pairs in the Lewis structure of SiO2. Hybridization Number = Number of atoms attached to the central atom + lone pairs on that central atom What is the concept of SiO2 hybridization? We must first determine its hybridization number. To determine the molecular geometry of SiO2, follow these steps. The VSEPR theory can be used to determine the molecular geometry of SiO2. SiO2 Molecular Geometry and Hybridization As a result, the valence shells of both silicon and oxygen have eight electrons. We convert two lone pairs of electrons from each oxygen atom to a covalent bond, as seen in the SiO2 Lewis dot structure. 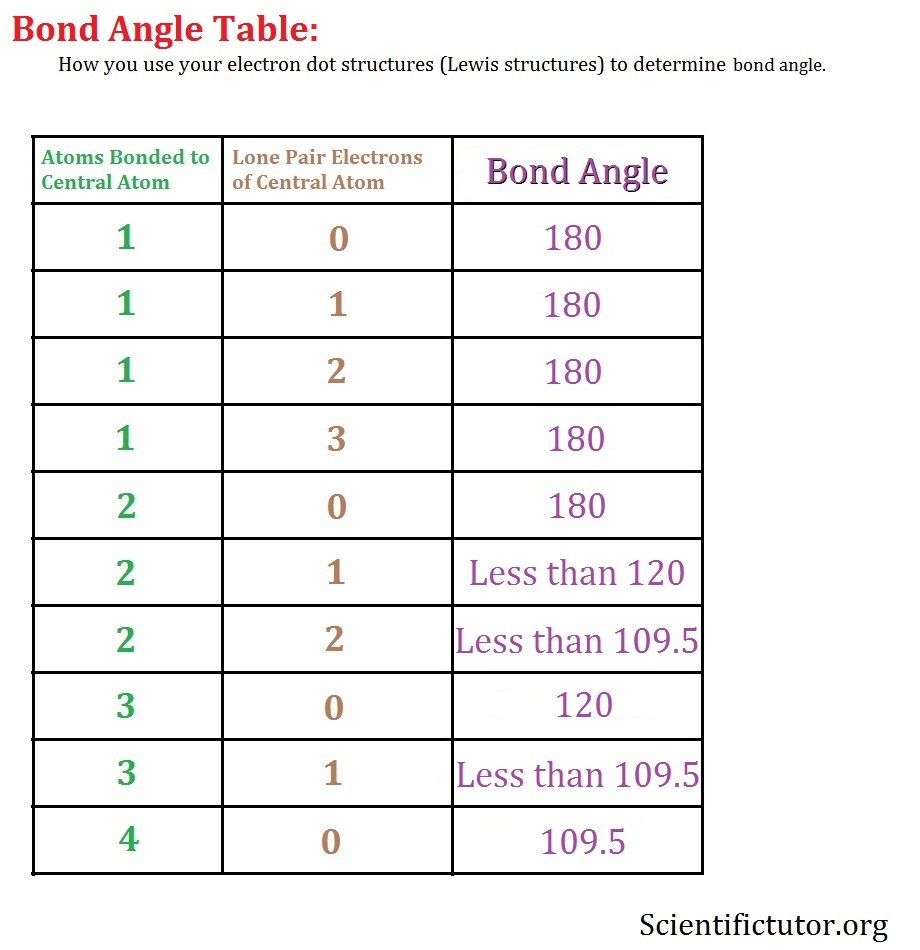
As we all know, silicon needs 8 electrons to complete its octet, but it only has 4 electrons right now.īy turning an oxygen atom’s electrons into a covalent bond, we would be able to achieve the octet of silicon. This is the last step in completing the Lewis diagram for SiO2. Step 5: Complete the central atom octet and, if possible, form a covalent bond. However, this structure is not yet stable since silicon only has four electrons surrounding it and requires four more to complete its octet. As a result, add six more electrons to each oxygen atom.įrom the total of 12 remaining valence electrons, we have put 6 electrons around each oxygen atom. However, oxygen already shares two electrons via a single bond. We need 12 more valence electrons to complete the octet, and oxygen needs 8 electrons. Position the remaining valence electron around the outer atom (oxygen) first. Now, we have an extra 12 valence electrons. Step 4: Starting with the outer atom, position the remaining valence electrons. It means that out of a total of 16 valence electrons, we used 4 of them. We used two single bonds in the structure since one single bond contains two electrons. Step 3: Using a single bond, connect all oxygen atoms to the silicon atom. So, place silicon in the middle, and oxygen is equally spaced around it. We learned that silicon is less electronegative than oxygen as electronegativity increases from left to right in the periodic table. We’ll look for the least electronegative atom between silicon and oxygen and place it in the center of the SiO2 Lewis structure. Step 2: Find the atom with the least electronegativity and position it in the middle. The total number of valence electrons available in the Lewis structure of SiO2 is 4 + 2*6 = 16 electrons. Silicon’s electronic configuration = 1s2 2s2 2p6 3s2 3p2 Oxygen’s electronic configuration = 1s2 2s2 2p4 Since silicon is in the 14th group of the periodic table and oxygen is in the 16th group, silicon has a 4 valence electron and oxygen has 6. 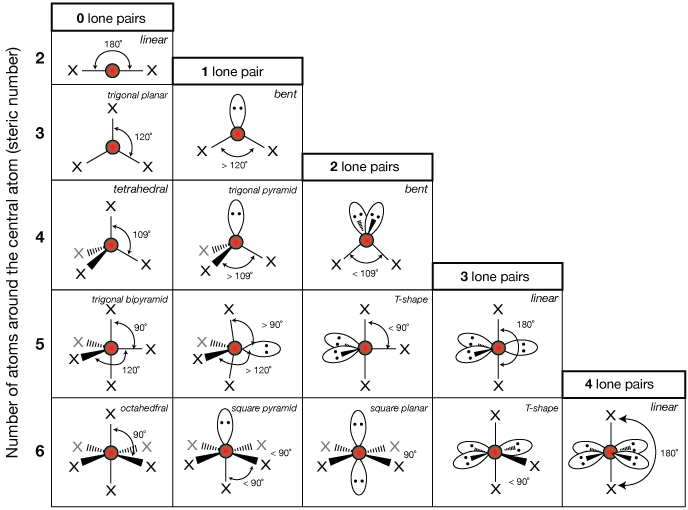
The first step is to determine how many valence electrons SiO2 has. Step 1: Count the total number of valence electrons in SiO2. Let’s take a look at how to do this step by step. There are no lone pairs on the central atom of the SiO2 Lewis dot structure, One silicon atom is at the middle, with two oxygen atoms bound to it in a double bond. The only difference is that instead of carbon, silicon is used. The Lewis structure of SiO2 is identical to the Lewis structure of CO2. Conclusion Silicon Dioxide (SiO2) Lewis Structure 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
